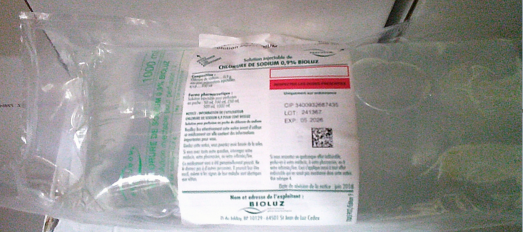
Sodium Chloride 0.9% Bioluz, Solution for Infusion in Dual Access PVC Bag 1000 mL (France)
Section 19A approved medicine
Sodium Chloride 0.9% Bioluz, Solution for Infusion in Dual Access PVC Bag 1000 mL (France)
Section 19A approval holder
Aborns Pharmaceuticals Pty Ltd ABN 80 625 808 193
Phone
1300 117 772
Approved until
Status
Current
Indication(s)
For extracellular fluid replacement and in the management of metabolic alkalosis in the presence of fluid loss, and for restoring or maintaining the concentration of sodium and chloride ions.
Images